.jpg)
Atoms of different elements have different properties and different atomic weights. Atoms of an element share common properties. Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen). What he learned led him to propose several laws, which are known collectively as Dalton's Atomic Theory or Dalton's Laws: Atoms are small, chemically indestructible particles of matter. The Law of Definite Proportions applies when elements are reacted together to form the same product. Any pure compound will always have the same ratio of the same elements. Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. This is the basic idea of the law of constant composition. Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. Atoms of different elements differ in size, mass, and other properties. Atoms of a given element are identical in size, mass, and other properties. Atoms cannot be subdivided, created, or destroyed. The general tenets of this theory are: All matter is composed of extremely small particles called atoms. If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C. Dalton proposed his atomic theory in 1804. It was 1913 before English physicist Henry Moseley reorganized the periodic table by atomic number.\): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B. He thought it was a question of inaccurate measurement or other experimental error. 
When he flipped his chart to a horizontal table two years later, he created a form much like what you see in chemistry textbooks and on the walls of chem labs today.Īlas, Mendeleev's table was based on atomic mass rather than atomic number, so details like the placement of tellurium and iodine didn't work out. Attempts to trace precisely how Dalton developed this theory have proved futile even Dalton’s own recollections on the subject are incomplete. 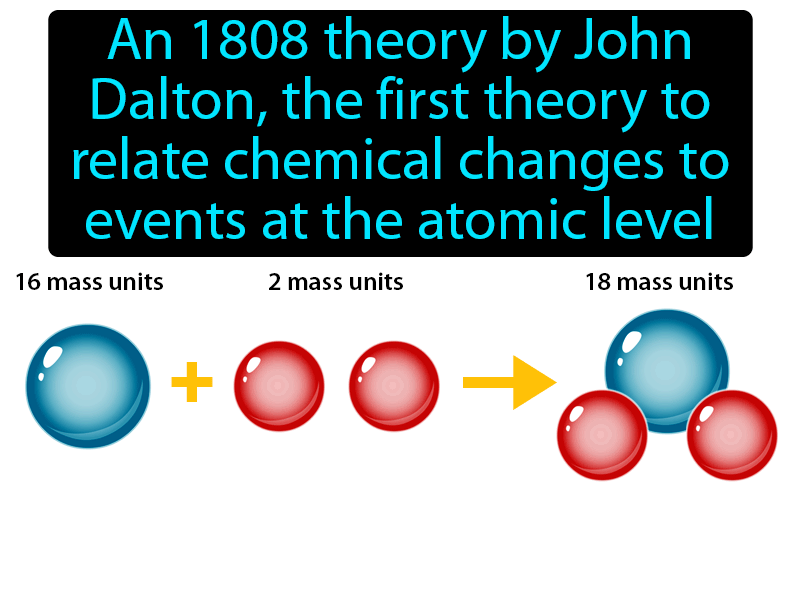 
The simplified notation led the way for English analytical chemist John Newlands to formulate his Law of Octaves and a prototype periodic table of the elements in 1864, but it was Russian chemist Dmitri Mendeleev who really laid it all on the table with 63 elements in 1869. John Dalton - Atomic Theory, Chemistry, Physics: By far Dalton’s most influential work in chemistry was his atomic theory. So it's Au for gold and Ag for silver, not the circled G and S of Dalton's original notation. In an era when all Europe's learned men (and the few women who were allowed into schools and universities) knew Latin, the shared language was an international lingua franca.Īll but a handful of Berzelius' symbols are still used today. Berzelius organized 47 elements with letters alone, and he based those letters not primarily on the English names, but on the Latin ones. Half of Dalton's symbols used letters inside a circle to represent the element. A decade after Dalton formulated his symbols, Swedish chemist Jöns Jakob Berzelius simplified the system. Dalton’s Atomic Theory It’s amazing to think about how life evolved on this planet.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
